Global warming has been attributed to the sharp increase in heat-trapping greenhouse gas emissions, in particular CO2 emissions. Carbon capture technology, such as using adsorbents to capture and store CO2 from ambient air, is a promising solution to mitigate emissions.
Liquid sorbents are traditionally used for carbon capture, but they suffer from equipment corrosion, high cost and high energy requirements for regeneration. To overcome these limitations, solid porous materials for CO2 adsorption—in which CO2 atoms adhere to the surface of the solid material—are being explored.
In his carbon capture research, Associate Professor Wu Ping of the Singapore University of Technology and Design (SUTD) turned to mica, a cheap and abundant clay mineral with diverse applications.
Mica forms sheet-like alumina silicate layers connected by interlayer potassium cations through ionic bonds. However, the complex structure makes it challenging to separate mica into single or few layers to form two-dimensional (2D) nanosheets that are conducive for CO2 capture. Methods developed by previous studies have also required long reaction times and high energy consumption.
To develop an efficient method to produce 2D mica nanosheets, Assoc Prof Wu and his SUTD team collaborated with researchers from the Agency for Science, Technology and Research (A*STAR). They published their research paper “Efficient synthesis of 2D mica nanosheets by solvothermal and microwave-assisted techniques for CO2 capture applications” in Materials.
“Building upon our recent breakthroughs in mechanochemistry, we have innovatively combined the techniques of microwave chemistry and solvothermal mechanochemistry. By harnessing the energy from microwaves and solvothermal processes, we were able to convert this energy into strain energy within solid-liquid-gas interfaces, facilitating the synthesis of exfoliated mica (eMica) nanosheets. These actions result in rapid exfoliation and significantly reduced reaction time,” explained Assoc Prof Wu.
The research team combined natural mica with potassium hydroxide in a polar solvent inside a closed reaction vessel. This reaction was then heated in a microwave, transferring energy to the microwave-absorbing polar solvent and the reactants. Coupled with the self-generated pressure inside the vessel, the mica was rapidly exfoliated with a significantly reduced reaction time. The microwave-treated mica was then sonicated to further expand and separate the layers. After several rounds of purification, the team had synthesized eMica nanosheets.
Compared to bulk mica, layers of eMica nanosheets are more uniform in lateral size and thickness. Furthermore, eMica nanosheets exhibit an ordered atomic arrangement, indicating their high quality and minimal defects.
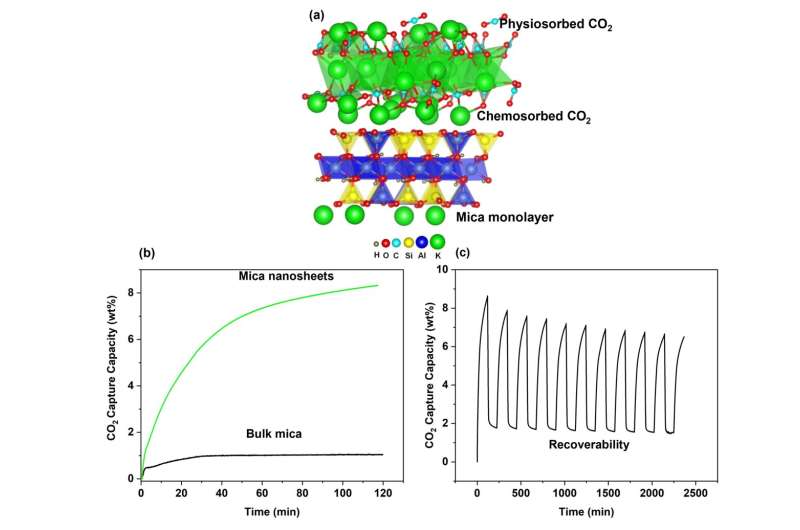
Associate Prof Wu and team next investigated the potential of the nanosheets for CO2 capture applications. They found that the CO2 adsorption capacity of eMica nanosheets was 87% higher than that of bulk mica. Though other types of adsorption materials in literature have demonstrated a higher capacity, eMica nanosheets still surpassed other clay minerals that have been modified for carbon capture.
The superior CO2 adsorption capacity of eMica nanosheets can be attributed to a high specific surface area and porosity between its expanded layers. The specific surface area of this 2D material had increased more than five times, from 29.1m2/g in bulk mica to 171.3m2/g in the nanosheets. The porosity of the nanosheets was also dramatically higher, with the pore volume increasing seven-fold, from 0.145cc/g in bulk mica to 1.022cc/g in eMica nanosheets.
CO2 adsorption could also have been boosted by deposits of potassium carbonate (K2CO3) on the nanosheets, which are formed when potassium cations in mica react with water and CO2 in the air. The team supported this hypothesis with computer simulations that demonstrated a K2CO3 deposited mica monolayer outperforming both bulk mica and a mica monolayer in CO2 adsorption.
Mechanistically, CO2 is captured by eMica nanosheets primarily through physical adsorption, forming weaker electrostatic attractions with the surface. This contrasts with the stronger ionic bonds that form when CO2 is chemically absorbed on the nanosheet surface, which occurs to a smaller extent. This predominant mechanism of physisorption would allow for easier CO2 desorption and the regeneration of the eMica nanosheets.
The research team found the nanosheets were able to maintain strong adsorption capacity when subjected to cyclic adsorption/desorption tests, demonstrating the recoverability and stability of the eMica nanosheets. Assoc Prof Wu believes this research will be of interest to the power generation sector, environmental and regulatory agencies and other researchers pursuing novel materials and technologies for CO2 capture. Additionally, his research contributes to the sustainability plans of SUTD.
“CO2 capture is an important aspect of mitigating greenhouse gas emissions, a key focus area for the sustainability strategy of SUTD. Our work on developing an efficient synthesis method aligns with the university’s emphasis on sustainable operations as well as sustainable education and research,” he commented.
Forging ahead, Assoc Prof Wu aims to develop a scalable method of mica exfoliation and explore the applications of mica for water purification.
“The scalable fabrication of 2D materials using sustainable and cost-effective methods could have significant implications for industry and society, such as reducing carbon emissions and improving energy efficiency. Overall, we hope that this research will advance our understanding of 2D materials and their potential applications and contribute to the development of sustainable and innovative technologies,” he said.
More information:
P. Vishakha T. Weerasinghe et al, Efficient Synthesis of 2D Mica Nanosheets by Solvothermal and Microwave-Assisted Techniques for CO2 Capture Applications, Materials (2023). DOI: 10.3390/ma16072921
Provided by
Singapore University of Technology and Design
Citation:
Scientists develop 2D nanosheets for sustainable carbon capture (2023, July 6)
retrieved 6 July 2023
from https://phys.org/news/2023-07-scientists-2d-nanosheets-sustainable-carbon.html
This document is subject to copyright. Apart from any fair dealing for the purpose of private study or research, no
part may be reproduced without the written permission. The content is provided for information purposes only.









